 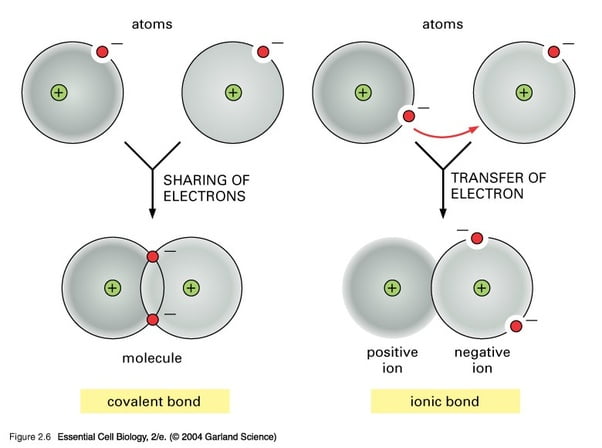
Chlorine has seven valence electrons in its outer orbit but to be in a stable condition, it needs eight electrons in outer orbit. Sodium and chlorine bonding ionically to form sodium chloride.įor example, in common table salt (NaCl) the individual atoms are sodium and chlorine. As long as the electro-negativity difference is no greater than 1.7, the atoms can only share the bonding electrons. The electro negativities (electron attracting ability) of the two bonded atoms are either equal or the difference is no greater than 1.7. This kind of bonds occurs mainly between a metallic and a non metallic atom.Ĭovalent bonds are formed as a result of the sharing of one or more pairs of bonding electrons. Ionic bond, also known as electrovalent bond, is a type of bond formed from the electrostatic attraction between oppositely charged ions in a chemical compound. So to stabilize the atom, each atom shares half of its electrons.Ĭovalent bonding is a form of chemical bonding between two non metallic atoms which is characterized by the sharing of pairs of electrons between atoms and other covalent bonds. But apart from Inert gas, this configuration is not present with most of the atoms. Each of these molecular orbits can have a certain number of electrons to form a stable atom. But electrons revolve in orbit around the center. At the centre of the atom, neutrons and protons stay together. The covalent bond is formed when two atoms are able to share electrons whereas the ionic bond is formed when the "sharing" is so unequal that an electron from atom A is completely lost to atom B, resulting in a pair of ions.Įach atom consists of protons, neutrons and electrons. Sodium chloride (NaCl), Sulphuric Acid (H2SO4 ) The two ions are attracted to each other and form an ionic bond. In ionic bonds, electrons are transferred from one atom to another, resulting in two charged ions - one positively charged, and one negatively charged. These two opposite ions attract each other and form the ionic bond. Non-metals(-ve ion) are "stronger" than the metal(+ve ion) and can get electrons very easily from the metal. 
For stabilization, they share their electrons from outer molecular orbit with others.Īn ionic bond is formed between a metal and a non-metal. 
Neither atom is "strong" enough to attract electrons from the other. These kinds of bonds occur mainly between a metallic and a non metallic atom.Ī covalent bond is formed between two non-metals that have similar electronegativities. Ionic bond, also known as electrovalent bond is a type of bond formed from the electrostatic attraction between oppositely charged ions in a chemical compound. Saturated Unsaturated and Supersaturatedĭuring the second world war, the American and British secret agencies came up with a so-called "L-pill," which could be given to operatives working beyond the front lines.Comparison chart Covalent Bonds versus Ionic Bonds comparison chartĬovalent bonding is a form of chemical bonding between two non metallic atoms which is characterized by the sharing of pairs of electrons between atoms and other covalent bonds. 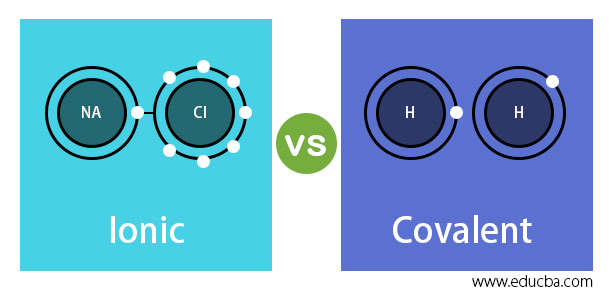
Reaction Quotient and Le Chatelier's Principle.Prediction of Element Properties Based on Periodic Trends.Molecular Structures of Acids and Bases.Ion and Atom Photoelectron Spectroscopy.Elemental Composition of Pure Substances.Application of Le Chatelier's Principle.Structure, Composition & Properties of Metals and Alloys.Intramolecular Force and Potential Energy.Variable Oxidation State of Transition Elements.Transition Metal Ions in Aqueous Solution.Single and Double Replacement Reactions. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
